Notes from the Field: Surveillance for Candida auris — Colombia, September 2016–May 2017
Weekly / April 20, 2018 / 67(15);459–460
Patricia Escandón1*; Diego H. Cáceres2,3*; Andres Espinosa-Bode4; Sandra Rivera1; Paige Armstrong2; Snigdha Vallabhaneni2; Elizabeth L. Berkow2; Shawn R. Lockhart2; Tom Chiller2; Brendan R. Jackson2; Carolina Duarte1 (View author affiliations)
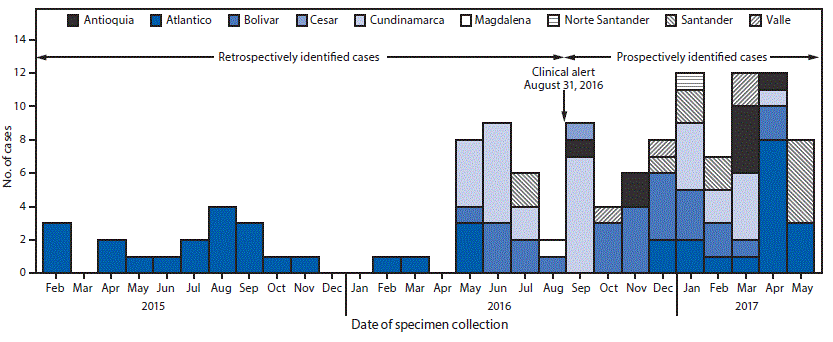
View suggested citationAfter a 2016 CDC alert describing infections caused by the multidrug-resistant fungus Candida auris (1), the Colombian Instituto Nacional de Salud (INS) queried the country’s WHONET† database of invasive Candida isolates to detect previous C. auris infections. No C. auris isolates were identified during 2012–2016. However, C. auris is often misidentified as Candida haemulonii (2), a yeast that rarely causes invasive infections, and 75 C. haemulonii isolates were reported during May 2013–August 2016. These isolates came primarily from patients in intensive care units in the country’s north region, approximately 350–600 km (220–375 miles) from Maracaibo, Venezuela, where C. auris cases were first identified in 2012 (3). Of the 75 reported Colombian C. haemulonii isolates in WHONET, INS obtained 45 isolates from six medical institutions dating from February 2015 through August 2016, all of which were confirmed to be C. auris by matrix-assisted laser desorption ionization-time of flight (MALDI-TOF) mass spectrometry. Based on these findings, INS issued a national alert and mandated reporting of suspected isolates on August 30, 2016§ (3,4). In September 2016, a team from INS, CDC, and medical staff members from hospitals with documented C. auris cases investigated the 45 MALDI-TOF–confirmed C. auris cases identified before the INS alert. This investigation involved two hospitals in the north region and two in the central region. Cases were clustered within specific hospital units, and surveillance sampling demonstrated transmission in health care settings (INS and CDC, unpublished data, 2018).
After release of the Colombian clinical alert, INS received suspected C. auris isolates for confirmatory testing, and during September 2016–May 2017, an additional 78 C. auris cases were identified from 24 health care facilities in nine states, resulting in a total of 123 confirmed C. auris cases (Figure), more than half (54.5%) recovered from the northern coastal region (Atlántico, Bolívar, and Cesar). The median age of all patients was 36 years (interquartile range = 2–62 years), and 75 (61%) were male. Children aged 0–18 years accounted for 39 (32%) cases, including 23 (19%) in infants aged <1 year. The majority (68; 56%) of cases were reported from the northern region, and 30 (24%) were reported from the central region. Isolates were recovered from blood (74; 60%), urine (11; 9%), respiratory specimens (10; 8%), the gastrointestinal tract (7; 5%), and other body fluids and body sites (8; 7%). For 13 (11%) cases, no information was available about the source of the C. auris isolate.
The VITEK 2 system had been used for yeast identification in 21 (75%) of 28 medical institutions. Four institutions used MicroScan (one), BD Phoenix (one), and Bruker MALDI-TOF Biotyper systems (two), and for three institutions, information about the identification method was not available. Six (4%) of 123 C. auris isolates were correctly identified, all by a clinical laboratory that used MALDI-TOF Biotyper (2). C. auris was most frequently misidentified as C. haemulonii (94; 76%), including 69 (97%) of 71 isolates identified by VITEK 2, all 23 isolates identified by BD Phoenix, and two of eight identified by MALDI-TOF Biotyper. Automated systems were unable to report a species for eight (7%) isolates (two by VITEK 2, four by MicroScan, and two by a system whose method was not reported). Thirteen C. auris isolates, all tested by MicroScan, were misidentified as other yeasts (Candida albicans, Candida guilliermondii, Candida parapsilosis, and Rhodotorula rubra).
Antifungal susceptibility testing was performed on 93 (76%) isolates¶ (2,5). Overall, 28 (30%) were resistant to fluconazole, 20 (22%) to amphotericin B, one (1%) to anidulafungin (an echinocandin), and one to both amphotericin B and anidulafungin.
Infections caused by C. auris are occurring in Colombia; the pathogen has been present in Columbia since at least 2015, and case counts are increasing. The number of reported cases likely does not reflect the true number of infected and colonized persons because of underreporting and underdiagnosis, as well as misdiagnosis as other yeast species (6). To contain the spread of C. auris in Colombia, INS updated the C. auris national clinical alert in July 2017 specifying which yeast isolates must be sent to INS for confirmation and mandating that medical facilities implement enhanced infection control practices, including using contact precautions and single rooms for patients with C. auris infections, minimizing the number of health care personnel in contact with infected patients, and daily and terminal cleaning of patient rooms and medical equipment with a disinfectant effective against Clostridium difficile spores** (2). Clinical laboratories should be aware that automated laboratory systems might incorrectly identify C. auris, particularly as C. haemulonii, although the species reported depends on the system (2).
Acknowledgments
Public Health Laboratories in Colombia; private laboratories; Unidad de Proteómica y Micosis Humana; Pontificia Universidad Javeriana; Indira Berrio, Corporacion para Investigaciones Biologicas (CIB), Medellin, Colombia; Maria Victoria Ovalle, INS; Anastasia Litvintseva, Nancy Chow, Cary Hilbert, Rory Welsh, Matthew Stuckey, Reina Turcios-Ruiz, Loren Cadena, Susan Kaydos-Daniels, Alex Bandea, Ngoc Le, Colleen Lysen, CDC.
Conflict of Interest
No conflicts of interest were reported.
Corresponding author: Patricia Escandón, pescandon@ins.gov.co, 57-1-220-7700, ext. 1420.
1Grupo de Microbiología, Instituto Nacional de Salud, Bogotá, Colombia; 2National Center for Emerging and Zoonotic Infectious Diseases, Office of Infectious Diseases, CDC; 3Oak Ridge Institute for Science and Education (ORISE), Oak Ridge, Tennessee; 4Division of Global Health Protection, Center for Global Health, CDC.
* These authors contributed equally.
† WHONET is a free software program developed by the World Health Organization (WHO) Collaborating Centre for Surveillance of Antimicrobial Resistance to support national surveillance activities in more than 120 countries (http://www.whonet.org/index.html).
§https://www.minsalud.gov.co/sites/rid/Lists/BibliotecaDigital/RIDE/IA/INS/ins-alerta-colombia-candida-auris.pdf.
¶ The broth microdilution method was used for azoles and echinocandins and Etest for amphotericin B; susceptibility breakpoints used were those described by CDC.
References
- CDC. Global emergence of invasive infections caused by the multidrug-resistant yeast. Atlanta, GA: US Department of Health and Human Services, CDC; 2016. https://www.cdc.gov/fungal/diseases/candidiasis/candida-auris-alert.html
- CDC. Recommendations for identification of Candida auris. Atlanta, GA: US Department of Health and Human Services, CDC; 2017. https://www.cdc.gov/fungal/diseases/candidiasis/recommendations.html
- Calvo B, Melo AS, Perozo-Mena A, et al. First report of Candida auris in America: clinical and microbiological aspects of 18 episodes of candidemia. J Infect 2016;73:369–74. CrossRefPubMed
- Chowdhary A, Sharma C, Duggal S, et al. New clonal strain of Candida auris, Delhi, India. Emerg Infect Dis 2013;19:1670–3. CrossRef PubMed
- Clinical and Laboratory Standards Institute. Reference method for broth dilution antifungal susceptibility testing of yeasts; approved standard. 3rd ed. Wayne, PA: Clinical and Laboratory Standards Institute; 2008.
- Morales-López SE, Parra-Giraldo CM, Ceballos-Garzón A, et al. Invasive infections with multidrug-resistant yeast Candida auris, Colombia. Emerg Infect Dis 2017;23:162–4. CrossRefPubMed






















.png)












No hay comentarios:
Publicar un comentario