


Safety Communication: FDA Recommends Against Use of Ovarian Cancer Screening TestsFDA is alerting women about the risks associated with the use of tests being marketed as ovarian cancer screening tests. The Agency is especially concerned about delaying effective preventive treatments for women who show no symptoms, but who are still at increased risk for developing ovarian cancer. Despite extensive research and published studies, there are currently no screening tests for ovarian cancer that are sensitive enough to reliably screen for ovarian cancer without a high number of inaccurate results. Based on FDA’s review of available clinical data from ovarian cancer screening trials and recommendations from healthcare professional societies and the U.S. Preventive Services Task Force, available data do not demonstrate that currently available ovarian cancer screening tests are accurate and reliable in screening asymptomatic women for early ovarian cancer. More information
Recall: Six batches of Novo Nordisk Inc. GlucaGen® HypoKit® (glucagon [rDNA origin] for injection) due to detached needles on the syringe in the kitNovo Nordisk Inc. is recalling six batches of the GlucaGen® HypoKit® in the U.S. due to two customer complaints from the UK and Portugal involving detached needles on the syringe with Sterile Water for Injection (SWFI). GlucaGen® HypoKit® is indicated for the treatment of severe hypoglycemia (low blood sugar) in patients with diabetes who are treated with insulin. A syringe with a detached needle cannot be used as prescribed. To protect patient safety, Novo Nordisk is recalling affected batches from wholesalers, pharmacies and patients in the U.S. It is estimated that out of the 71,215 pens being recalled, four pens could be defective. More information
|

Recall: Virtus Pharmaceuticals Opco II, LLC ofHyoscyamine sulfate Due to Superpotent and Subpotent Results
Virtus Pharmaceuticals Opco II, LLC (Virtus) is voluntarily recalling seven batches of Hyoscyamine sulfate (0.125mg) to the consumer level which include the tablet, sublingual, and orally disintegrating tablet form. Hyoscyamine sulfate is an anticholinergic agent which blocks the action of acetylcholine and is used to treat diseases like asthma, incontinence, stomach cramps, peptic ulcers, control gastric secretion, intestinal spasm and other bowel disturbances. This recall is being initiated due to both superpotent and subpotent test results. All of these batches were manufactured by Pharmatech LLC for distribution by Virtus throughout the United States and Puerto Rico. Taking a product that is superpotent could result in hot/dry skin, fever, blurred vision, sensitivity to light, dry mouth, unusual excitement, fast or irregular heartbeat, dizziness, an inability to completely empty the bladder, and seizures. The severity of the adverse event would depend on how superpotent the tablet was. Adverse events such as clotted blood within the tissues and fractures could occur, as a result of falls from dizziness or seizures if the strength is particularly high.More information |
Comunicaciones de la FDA sobre la seguridad de los medicamentos en español
Descargo de responsabilidad: La FDA reconoce la necesidad de proporcionar información importante sobre seguridad de los medicamentos en idiomas distintos al inglés. Hacemos lo mejor posible para proporcionar versiones en español precisas y oportunas de nuestras Comunicaciones de Seguridad de Medicamentos. Sin embargo, en caso que existiera discrepancias entre las versiones en inglés y la de español, la información contenida en la versión en inglés es la que se considera como versión oficial. Si tiene alguna pregunta, por favor contáctese con Division of Drug Information en druginfo@fda.hhs.gov.Comunicaciones de la FDA |

FDA knows the major public health consequences that can result from drug shortages. These shortages occur for many reasons including manufacturing and quality problems, delays and discontinuations. When issues are discovered, FDA works closely with the company to address risks involved to prevent harm to patients. FDA also considers the impact a shortage would have on patient care and access and works with the company to restore supplies while also ensuring safety for patients. More information
|
Drug Shortages Voluntarily Reported by Manufactures During the Past 2 Weeks:
- Etoposide Phosphate (Etopophos) Injection
- Scopolamine (Transderm Scop) Transdermal System Patch
- Water-Miscible Vitamin A Palmitate (Aquasol A Parenteral)
Drug Shortages Reported to be Resolved by Manufacturers During the Past 2 Weeks:
Drug Discontinuation Voluntarily Reported by Manufacturers During the Past 2 Weeks:
Information about blood and biologic shortages, resolved shortages, and discontinuations
La FDA reconoce las consecuencias significativas para la salud pública que pueden resultar de la escasez de medicamentos y hace un gran esfuerzo dentro de sus facultades legales para abordar yprevenir la escasez de medicamentos. La escasez se produce por muchas razones, incluyendoproblemas de fabricación y calidad, retrasos y discontinuación del producto. Cuando los problemas son descubiertos por la empresa o el público y reportados a la FDA o se descubren por inspecciones de la FDA, la FDA trabaja en estrecha colaboración con la empresa para hacer frente a los riesgosinvolucrados y evitar daños a los pacientes. La FDA también considera el impacto que una escaseztendría en la atención médica del paciente y al acceso del producto y trabaja con la empresa pararestablecer el suministro al tiempo que garantiza la seguridad de los pacientes. Más información

FDA grants accelerated approval to first drug for Duchenne muscular dystrophyFDA has approved Exondys 51 (eteplirsen) injection, the first drug approved to treat patients with Duchenne muscular dystrophy (DMD). Exondys 51 is specifically indicated for patients who have a confirmed mutation of the dystrophin gene amenable to exon 51 skipping, which affects about 13 percent of the population with DMD. “Patients with a particular type of Duchenne muscular dystrophy will now have access to an approved treatment for this rare and devastating disease,” said Janet Woodcock, M.D., director of the FDA’s Center for Drug Evaluation and Research. “In rare diseases, new drug development is especially challenging due to the small numbers of people affected by each disease and the lack of medical understanding of many disorders. Accelerated approval makes this drug available to patients based on initial data, but we eagerly await learning more about the efficacy of this drug through a confirmatory clinical trial that the company must conduct after approval.”More information
FDA permits marketing of balloon device to treat persistent Eustachian tube dysfunctionFDA is permitting marketing of a device that uses a small balloon to treat persistent Eustachian tube dysfunction (ETD), a condition in which pressure, pain or clogged or muffled sensations occur in the ear. The Eustachian tube connects the middle ear to the back of the throat. It is normally filled with air and helps maintain equal pressure inside the ear with the surrounding environment by periodically opening and closing, like a valve. If this function is impaired, as is the case with ETD, it results in discomfort, impaired hearing, persistent ear infections, ringing in the ears (tinnitus) or other symptoms. More information
|
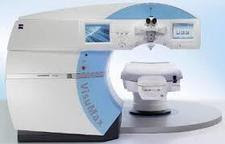
FDA approves VisuMax Femtosecond Laser to surgically treat nearsightednessFDA has approved the VisuMax Femtosecond Laser for the small incision lenticule extraction (SMILE) procedure to reduce or eliminate nearsightedness in certain patients 22 years of age or older. Not all patients are candidates for SMILE, and individuals should carefully review the patient labeling and discuss their expectations with their eye care professional. “This approval expands the surgical treatment options available to patients for correcting nearsightedness,” said Malvina Eydelman, M.D., director of Ophthalmic and Ear, Nose and Throat Devices, in FDA’s Center for Devices and Radiological Health. More information
FDA approves Amjevita, a biosimilar to HumiraFDA has approved Amjevita (adalimumab-atto) as a biosimilar to Humira (adalimumab) for multiple inflammatory diseases. Amjevita is also indicated for moderately to severely active polyarticular juvenile idiopathic arthritis in patients four years of age and older. “This is the fourth FDA-approved biosimilar. The biosimilar pathway is still a new frontier and one that we expect will enhance access to treatment for patients with serious medical conditions,” said Janet Woodcock, M.D., director of the FDA’s Center for Drug Evaluation and Research. More information
FDA approves expanded indications for Ilaris for three rare diseases
FDA has approved three new indications for Ilaris (canakinumab). The new indications are for rare and serious auto-inflammatory diseases in adult and pediatric patients: Tumor Necrosis Factor Receptor Associated Periodic Syndrome (TRAPS), Hyperimmunoglobulin D Syndrome (HIDS)/Mevalonate Kinase Deficiency (MKD), and Familial Mediterranean Fever (FMF). All three syndromes are hereditary diseases that are characterized by periodic attacks of fever and inflammation, as well as severe muscle pain. There are no previously approved therapies for TRAPS or HIDS/MKD. More information |
For information on drug approvals or to view prescribing information and patient information, please visit Drugs@FDA or DailyMed.

View FDA's Comments on Current Draft Guidance page, for a list of current draft guidances and other topics of interest for patients and caregivers.
|

FDA launches competition to spur innovative technologies to help reduce opioid overdose deathsFDA has announced the 2016 Naloxone App Competition, a public contest focused on developing innovative technologies to combat the rising epidemic of opioid overdose. FDA, with support from the National Institute on Drug Abuse (NIDA) and the Substance Abuse and Mental Health Services Administration (SAMHSA), is inviting computer programmers, public health advocates, clinical researchers, entrepreneurs and innovators from all disciplines to create a mobile phone application that can connect opioid users experiencing an overdose with nearby carriers of the prescription drug naloxone – the antidote for an opioid overdose – thereby increasing the likelihood of timely administration and overdose reversal. This competition builds on work announced in the FDA’s Opioids Action Plan and the U.S. Department of Health and Human Services’ Opioid Initiative to take concrete steps toward reducing the impact of opioid misuse, dependence and overdose on American families and communities by making naloxone more accessible. More information
|

Evaluating FDA’s Approach to Cancer Clinical Trials, by Richard Pazdur, M.D., FDA’s Acting Director, Oncology Center of ExcellenceSince the announcement of the FDA Oncology Center of Excellence (OCE) two months ago (June 29, 2016) as part of the White House’s Cancer Moonshot, we’ve been working to further FDA’s efforts to get new oncology products into the hands of patients. We are committed to meet the needs of patients and health care communities by driving progress in the prevention, diagnosis, and treatment of cancer. At the core of the OCE’s work – and of the Cancer Moonshot – is taking a new look at what we have been doing in the past so we can operate more efficiently in the future. The OCE will leverage the combined skills of oncologists and scientists with expertise in drugs, biologics, and devices to employ the best and most innovative approaches to bring forth safe new oncology products.
To read the rest of this post, see FDA Voice Blog, September 16, 2016.
FDA Warns Consumers of Extortion Scam
Since 2008, hundreds of people who have purchased prescription drugs over the Internet or via telephone have unknowingly exposed themselves to extortion by individuals posing as FDA and DEA agents. FDA Drug Info Rounds pharmacists discuss some simple steps to take if a potential victim receives this type of call. Video / Transcript / Other FDA Drug Info Rounds Videos
Since 2008, hundreds of people who have purchased prescription drugs over the Internet or via telephone have unknowingly exposed themselves to extortion by individuals posing as FDA and DEA agents. FDA Drug Info Rounds pharmacists discuss some simple steps to take if a potential victim receives this type of call. Video / Transcript / Other FDA Drug Info Rounds Videos
FDA Issues Report from the Patient-Focused Drug Development for Alpha-1 Antitrypsin Deficiency Meeting
On September 29, 2015, FDA held a public meeting to hear perspectives from patients with Alpha-1 Antitrypsin Deficiency (AATD), their caregivers, and other patient representatives. The meeting enabled discussion on the impact that AATD has on patients’ daily lives, as well as discussions on currently available therapies, and patient considerations on drug development. FDA conducted the meeting as part of the Agency’s Patient-Focused Drug Development initiative, an FDA commitment under the fifth authorization of the Prescription Drug User Fee Act (PDUFA V) to more systematically gather patients’ perspectives on their condition and on therapies that are available to treat their condition. The Report
On September 29, 2015, FDA held a public meeting to hear perspectives from patients with Alpha-1 Antitrypsin Deficiency (AATD), their caregivers, and other patient representatives. The meeting enabled discussion on the impact that AATD has on patients’ daily lives, as well as discussions on currently available therapies, and patient considerations on drug development. FDA conducted the meeting as part of the Agency’s Patient-Focused Drug Development initiative, an FDA commitment under the fifth authorization of the Prescription Drug User Fee Act (PDUFA V) to more systematically gather patients’ perspectives on their condition and on therapies that are available to treat their condition. The Report

In this section you will find a comprehensive list of all the meetings that the FDA is involved with. The meetings may include advisory committee meetings, public workshops and public conferences that are seeking to hear from patients and caregivers.
Most FDA meetings are free to the public and do not require the public to register. Interested persons may present data, information, or views, orally at the meeting, or in writing, on issues pending before the committee. Other types of meetings listed may require prior registration and fees.
|
View FDA's Patient Network Calendar of Public Meetings page for a complete list of meetings and workshops.
For additional information on other agency meetings please visit Meetings, Conferences, & Workshops.
|

Medication Safety Tips for Older Adults
Whether you’re settling into your sixties or heading into your ninth decade, be careful when taking prescription and over-the-counter medicines, herbal preparations, and supplements. And if you’re caring for older loved ones, help them stay safe, too. Why the special concern? The older you get, the more likely you are to use additional medicines, which can increase the chance of harmful drug effects, including interactions. And, as you age, physical changes can affect the way medicines are handled by your body, leading to potential complications. For instance, your liver and kidneys may not work as well, which affects how a drug breaks down and leaves your body. More information
FDA: Cutting-Edge Technology Sheds Light on Antibiotic Resistance
When your child has a bacterial infection, you take her to the doctor, get a prescription for an antibiotic, and, in most cases, all is soon well. But while antibiotics work for most patients with a bacterial infection, they may not for all infections. Public health agencies around the world are dealing with the growing challenge of bacterial resistance to antibiotics, which can make these medications ineffective. The Centers for Disease Control and Prevention (CDC) reports that every year at least 2 million illnesses and 23,000 deaths in the United States are caused by antibiotic-resistant bacteria. The problem is especially serious for patients who have few viable antibiotic options; this can occur with certain types of serious bloodstream infections and with gonorrhea infections, among others. More information |
More Consumer Updates
For previously published Consumer Update articles that are timely and easy-to-read and cover all FDA activities and regulated products. More information
For previously published Consumer Update articles that are timely and easy-to-read and cover all FDA activities and regulated products. More information
En Español
La información en esta página es para el público en general, y para profesionales y educadores de salud. Esta información puede ser distribuida y publicada sin previa autorización. En Español
La información en esta página es para el público en general, y para profesionales y educadores de salud. Esta información puede ser distribuida y publicada sin previa autorización. En Español

Animal Health LiteracyAnimal Health Literacy means timely information for the benefit of all animals and their humans. With continuous communication and outreach, the Center for Veterinary Medicine (CVM) strives to enhance the public trust, promote safe and effective use of the animal health products we regulate, and share our scientific endeavors. CVM provides reliable, science-based information to promote animal and human health. More information / Publicaciones en Español del FDA
Animal and Veterinary UpdatesAnimal and veterinary updates provide information to keep your pets healthy and safe. More information
|

How to Report a Pet Food Complaint
You can report complaints about a pet food product electronically through the Safety Reporting Portal or you can call your state’s FDA Consumer Complaint Coordinators. Please provide as much information as possible in your complaint, such as exact name of product, type of container, lot number, UPC codes, how the food was stored, and purchase date and exact location where purchased. If possible, please save the original packaging until the pet food has been consumed. The packaging contains IMPORTANT information often needed to identify the variety of pet food, the manufacturing plant, and the production date. More information |

Animal Health LiteracyAnimal Health Literacy means timely information for the benefit of all animals and their humans. With continuous communication and outreach, the Center for Veterinary Medicine (CVM) strives to enhance the public trust, promote safe and effective use of the animal health products we regulate, and share our scientific endeavors. CVM provides reliable, science-based information to promote animal and human health. More information / Publicaciones en Español del FDA
Animal and Veterinary UpdatesAnimal and veterinary updates provide information to keep your pets healthy and safe. More information
|

How to Report a Pet Food Complaint
You can report complaints about a pet food product electronically through the Safety Reporting Portal or you can call your state’s FDA Consumer Complaint Coordinators. Please provide as much information as possible in your complaint, such as exact name of product, type of container, lot number, UPC codes, how the food was stored, and purchase date and exact location where purchased. If possible, please save the original packaging until the pet food has been consumed. The packaging contains IMPORTANT information often needed to identify the variety of pet food, the manufacturing plant, and the production date. More information |

FDA protects kids from illegal sales of e-cigarettes, e-liquids and cigars
FDA has announced it has taken action against 55 tobacco retailers by issuing the first warning letters for selling newly regulated tobacco products, such as e-cigarettes, e-liquids and cigars, to minors. During compliance checks at major national retail chains, tobacco specialty stores and online retailers, minors were able to purchase some of these newly regulated tobacco products in a variety of youth-appealing flavors, including bubble gum, cotton candy and gummy bear. More information |

How Do FDA's New Regulations for E-Cigarettes, Cigars, and All Other Tobacco Products Affect Consumers?
Tobacco use is the single largest preventable cause of disease and death in the United States. As part of its goal to improve public health and protect future generations from the risks of tobacco use, FDA has extended its authority to cover all tobacco products. The fact that FDA regulates these tobacco products does not mean they are safe to use. More information
Public Health Education
Tobacco products are harmful, yet widely used, consumer products that are responsible for severe health problems in both users and non-users. These health problems include cancer, lung disease, and heart disease, which often lead to death.
|
Public Education CampaignsWe are investing in a number of public education campaigns, such as Fresh Empire and The Real Cost, to help educate the public – especially youth – about the dangers of regulated tobacco products. Rooted in science, these efforts are directly linked to our authority to regulate the marketing and sales of tobacco products. More information
Youth and TobaccoWe are working to protect the health of America’s children and ultimately reduce the burden of illness and death caused by tobacco use. More information

What is a Cosmetic?The Federal Food, Drug, and Cosmetic Act defines cosmetics by their intended use, as "articles intended to be rubbed, poured, sprinkled, or sprayed on, introduced into, or otherwise applied to the human body...for cleansing, beautifying, promoting attractiveness, or altering the appearance". Among the products included in this definition are skin moisturizers, perfumes, lipsticks, fingernail polishes, eye and facial makeup preparations, cleansing shampoos, permanent waves, hair colors, and deodorants, as well as any substance intended for use as a component of a cosmetic product. More information
|
Recalls and Alerts
To see safety alerts and recent recalls related to cosmetics and other products regulated by FDA. More information
To see safety alerts and recent recalls related to cosmetics and other products regulated by FDA. More information
How to report a problemIn the case of a reaction or problem with a cosmetic product, the first step is to stop using the product and contact your healthcare provider. FDA does not provide medical advice. Then, report the problem to FDA. You can report a rash, redness, burn, hair loss, headache, infection, illness, or any other unexpected reaction, whether or not it required medical treatment. You can also report a bad smell, color change, or other sign of contamination. You can choose the way you’d prefer to report:
- Call an FDA Consumer Complaint Coordinator if you want to speak directly to a person about your problem.
- Complete an electronic Voluntary MedWatch form online.
- Complete a paper Voluntary MedWatch form that you can mail to FDA.

Information about Expanded Access
Expanded access, sometimes called "compassionate use," is the use outside of a clinical trial of an investigational medical product (i.e., one that has not been approved by FDA). FDA is committed to increasing awareness of and knowledge about its expanded access programs and the procedures for obtaining access to human investigational drugs (including biologics) and medical devices. More information |
Learn about what your physician should do before submitting a request for individual patient expanded access use of an investigational medical product, who may be eligible for expanded access, associated costs, FDA contacts and more. Information for Patients
|
Learn about your responsibilities under the expanded access pathway, how to submit a request for expanded access for an individual patient (including for emergency use), which forms to use, FDA contacts and more. Information for Physicians
|

FDA Patient Network
The FDA Patient Network contains a series of webpages, webinars and presentations on topics related to patient engagement, FDA regulations, understanding medical product (Drugs, Biologics, and Devices) approval and medical product safety updates, Take me to the FDA Patient Network or take me to FDA Webinars.
FDA Basics
Each month, different centers and offices at FDA will host an online session where the public can ask questions to senior FDA officials about a specific topic or just listen in to learn more about FDA. More information |

healthfinder.gov
Welcome to healthfinder.gov, a government Web site where you will find information and tools to help you and those you care about stay healthy.More information / más información
FDA Email Updates
Sign up for one of the FDA disease specific e-mail list that delivers updates, including product approvals, safety warnings, notices of upcoming meetings, and notices on proposed regulatory guidances. |






















.png)












No hay comentarios:
Publicar un comentario