 |
| MMWR Weekly Vol. 65, No. 48 December 09, 2016 |
| PDF of this issue |
CDC Grand Rounds: Modeling and Public Health Decision-Making
Weekly / December 9, 2016 / 65(48);1374–1377
Leah S. Fischer, PhD1; Scott Santibanez, MD1; Richard J. Hatchett, MD2; Daniel B. Jernigan, MD3; Lauren Ancel Meyers, PhD4; Phoebe G. Thorpe, MD5; Martin I. Meltzer, PhD1 (View author affiliations)
View suggested citation
Mathematical models incorporate various data sources and advanced computational techniques to portray real-world disease transmission and translate the basic science of infectious diseases into decision-support tools for public health. Unlike standard epidemiologic methods that rely on complete data, modeling is needed when there are gaps in data. By combining diverse data sources, models can fill gaps when critical decisions must be made using incomplete or limited information. They can be used to assess the effect and feasibility of different scenarios and provide insight into the emergence, spread, and control of disease. During the past decade, models have been used to predict the likelihood and magnitude of infectious disease outbreaks, inform emergency response activities in real time (1), and develop plans and preparedness strategies for future events, the latter of which proved invaluable during outbreaks such as severe acute respiratory syndrome and pandemic influenza (2–6). Ideally, modeling is a multistep process that involves communication between modelers and decision-makers, allowing them to gain a mutual understanding of the problem to be addressed, the type of estimates that can be reliably generated, and the limitations of the data. As models become more detailed and relevant to real-time threats, the importance of modeling in public health decision-making continues to grow.
Predicting the Likelihood, Timing, and Magnitude of Infectious Disease Outbreaks
Federal agencies and academic partners are working to produce models with short- and long-term projections of when and where outbreaks will occur (7). For example, the “Predict the Influenza Season” challenge, started in 2013, moved influenza forecasting forward by engaging the scientific community to develop innovative and cost-effective methods to predict influenza activity and to more clearly identify areas of uncertainty in forecasting flu activity (8). This ongoing project encourages participants to predict the timing, peak, and intensity of influenza seasons by combining social media data (e.g., Twitter, internet search data, web surveys, etc.) and data from CDC’s routine influenza surveillance systems (9). As part of the Influenza Virologic Surveillance Right Size project, a public health-academic partnership developed models that determine the minimum weekly number of specimens to be screened per public health laboratory to efficiently detect emerging viruses and select strains for inclusion in the next seasonal influenza vaccine (10).
Providing Real-Time Insight During Public Health Emergencies
During public health emergencies, decision-makers need to quantify the risk to the public, delineate priorities with a clear and narrow focus, and maintain flexibility in considering options. During outbreak responses, modelers are asked to estimate the size of populations at risk for disease or death and the potential impact of interventions on both the timing and public health burden of an outbreak (Figure). By facilitating dialogue about what data are available and what data are needed to answer these questions, modelers can aid decision-makers as an outbreak situation evolves (11). Framing and addressing such questions via models helps leadership understand the appropriate size, type, time frame, and scale of resources needed to deploy interventions to maximize their impact. For example, one model produced during the Ebola virus disease (Ebola) response predicted the likelihood of the spread of Ebola from districts with reported Ebola cases to specific districts and neighboring countries with no reported cases. This forecast of geographic spread of Ebola allowed decision-makers to prioritize where to direct resources to improve surveillance (12).
To provide insight, modelers often must extract and combine useful information from diverse data sources, including traditional surveillance data, laboratory data, and social media, and collate them into meaningful information. Early in the West African Ebola epidemic, researchers at the University of Texas at Austin and Yale University used a combination of viral sequence data and case counts reported on the Sierra Leone Ministry of Health Facebook page to estimate the rate of spread and the clustered nature of Ebola transmission (13). During the 2009 H1N1 influenza pandemic, CDC modelers provided leaders, policy makers, and the public with near real-time modeled estimates of cases, hospitalizations, and deaths, corrected for underreporting (14,15). Before sufficient epidemiologic data existed, the modeled data allowed public health officials to more readily appreciate the magnitude of the disease transmission and understand the dynamic of the pandemic as risk patterns changed over time. Knowing where influenza is spreading in near real time and anticipating the timing and severity of the peak can improve clinical practice by facilitating plans for hospital and laboratory surge capacity and the implementation of pharmaceutical and nonpharmaceutical interventions (16). This insight gives decision-makers more flexibility to match resources to needs during public health emergencies.
Modelers have provided critical support for emergency response activities by estimating the size and potential growth of outbreaks before large amounts of data were available, assessing the potential impact of interventions, identifying important data needs (e.g., value of what is known, value of what is not known, prioritization of data collection), and developing simple decision-support tools for broad dissemination (11)
Looking Back to Plan Ahead
Models can improve preparedness planning for infectious disease outbreaks and emergencies by providing critical information for quantitative public health decisions, such as those related to stockpiling and allocating public health resources and medical countermeasures (17). For example, during the Ebola response, modelers developed a tool to estimate the resources that might have been needed at any one time to treat Ebola patients if Ebola became widespread in the United States (18). Modelers also produced estimates to answer many post-H1N1 pandemic questions, including which groups experienced the most risk. Building on the modeling developed during that pandemic, modelers were able to confirm that influenza-related deaths and hospitalizations in children aged < 18 years reached pandemic levels, when compared with influenza-related deaths during nonpandemic influenza seasons (14). Other valuable data estimating the number of cases, hospitalizations, and deaths averted because of vaccination and the use of influenza antiviral drugs can be used to allow public health officials to prepare for the next influenza pandemic.
Comparing model predictions with observations of real-life events can yield improvements in both model structure and parameter estimates. In this way, models “learn” from past outbreaks to improve data collection, situational awareness, and outbreak prediction. For example, after modelers assessed the effects of the 2009–2010 H1N1 influenza virus vaccination program by estimating the number of clinical cases, hospitalizations, and deaths prevented (19,20), CDC implemented a standardized data set and annual assessment of estimated seasonal influenza illnesses and hospitalizations averted because of vaccination, which can be used to improve model predictions.
Facilitating Communication
In addition to offering insight, modeling can assist communication among the multiple decision-makers involved with public health emergencies. Because models should only be used for the purpose for which they were intended, the back-and-forth dialogue required to ensure decision-makers understand the limitations of a specific model creates opportunities for leaders to articulate public health goals and better understand factors contributing to the dynamics of the modeled outbreak. These dialogues also allow decision-makers to explore the feasibility of interventions and estimate the resources required to implement such options.
Challenges and Limitations
A number of challenges can occur with the use of models for emergency response, planning, and preparedness. The technical challenges modelers typically encounter include a lack of quality and real-time data. Many models, such as those that predicted case counts of Ebola, are developed for a specific purpose, and thus might not be necessary for future planning. However, models that can be used repeatedly over time need clear plans for maintenance and future availability (20). The continued relevance and utility of models also can be impeded by evolving operating systems, web software, format of data inputs, and practical requirements for direct manipulation by model developers.
Other challenges that modelers and decision-makers confront relate to a lack of understanding about the modeling process and its limitations. The modeling process relies upon the questions that direct the development of estimates and projections produced (Box). An awareness of these guiding questions helps decision-makers better interpret and understand the limitations of models.
Models can help frame decision choices, but will seldom tell decision-makers which specific choices to make. Like every other tool, models can be misused, intentionally or unintentionally. Models are stylized representations of the world operating under specific assumptions; therefore, models capture only a part of the world’s complexity. Decision-makers should be careful not to draw conclusions outside of the problem areas the model was designed to address.
Looking Ahead
Mathematical models are valuable decision-support tools that reveal outbreak dynamics, improve planning and preparedness, and aid communication between modelers and decision-makers. Future modeling possibilities are broad; for example, real-time genomic and antigenic virus fitness forecasting for selection of the best vaccine virus candidates is a possibility. As data availability and the accuracy of predictions improve, models will continue to provide valuable information to guide public health decision-makers. However, to sustain and advance modeling, attention and resources must be dedicated to improving data access, codifying best practices, and improving the nation’s capacity to do modeling work. Modeling serves as an increasingly valuable resource for decision-makers in the emergence, spread, and control of outbreaks, and continued investments will pay large dividends over the long-term.
Corresponding author: Leah S. Fischer, lsfischer@cdc.gov, 404-639-4227.
1Division of Preparedness and Emerging Infections, National Center for Emerging and Zoonotic Infectious Diseases, CDC; 2Biomedical Advanced Research and Development Authority, Office of the Assistant Secretary for Preparedness and Response, U.S. Department of Health and Human Services; 3Influenza Division, National Center for Immunization and Respiratory Disease, CDC; 4Department of Integrative Biology, University of Texas at Austin; 5Office for the Associate Director of Science, CDC.
References
- Meltzer MI, Atkins CY, Santibanez S, et al. Estimating the future number of cases in the Ebola epidemic—Liberia and Sierra Leone, 2014–2015 . MMWR Surveill Summ 2014;63
- Fraser C, Riley S, Anderson RM, Ferguson NM. Factors that make an infectious disease outbreak controllable. Proc Natl Acad Sci U S A 2004;101:6146–51. CrossRef PubMed
- Meyers LA, Pourbohloul B, Newman MEJ, Skowronski DM, Brunham RC. Network theory and SARS: predicting outbreak diversity. J Theor Biol 2005;232:71–81. CrossRef PubMed
- Dimitrov NB, Goll S, Hupert N, Pourbohloul B, Meyers LA. Optimizing tactics for use of the U.S. antiviral strategic national stockpile for pandemic influenza. PLoS One 2011;6:e16094.CrossRef PubMed
- Lipsitch M, Finelli L, Heffernan RT, Leung GM, Redd SC; 2009 H1n1 Surveillance Group. Improving the evidence base for decision making during a pandemic: the example of 2009 influenza A/H1N1. Biosecur Bioterror 2011;9:89–115. PubMed
- Glasser JW, Hupert N, McCauley MM, Hatchett R. Modeling and public health emergency responses: lessons from SARS. Epidemics 2011;3:32–7. CrossRef PubMed
- Cauchemez S, Epperson S, Biggerstaff M, Swerdlow D, Finelli L, Ferguson NM. Using routine surveillance data to estimate the epidemic potential of emerging zoonoses: application to the emergence of US swine origin influenza A H3N2v virus. PLoS Med 2013;10:e1001399. CrossRef PubMed
- Biggerstaff M, Alper D, Dredze M, et al. ; Influenza Forecasting Contest Working Group. Results from the centers for disease control and prevention’s predict the 2013-2014 Influenza Season Challenge. BMC Infect Dis 2016;16:357. CrossRef PubMed
- CDC. CDC announces winner of the ‘Predict the Influenza Season Challenge.’ Atlanta, GA: US Department of Health and Human Services, CDC; 2015. http://www.cdc.gov/flu/news/predict-flu-challenge-winner.htm
- Association of Public Health Laboratories. Influenza virologic surveillance right size roadmap. Silver Spring, MD: Association of Public Health Laboratories; 2013. https://www.aphl.org/AboutAPHL/publications/Documents/ID_July2013_Influenza-Virologic-Surveillance-Right-Size-Roadmap.pdf
- Meltzer MI, Santibanez S, Fischer LS, et al. Modeling in real time during the Ebola response. MMWR Suppl 2016;65:85–9. PubMed
- Rainisch G, Shankar M, Wellman M, Merlin T, Meltzer MI. Regional spread of Ebola virus, West Africa, 2014. Emerg Infect Dis 2015;21:444–7. CrossRef PubMed
- Scarpino SV, Iamarino A, Wells C, et al. Epidemiological and viral genomic sequence analysis of the 2014 Ebola outbreak reveals clustered transmission. Clin Infect Dis 2015;60:1079–82. PubMed
- Shrestha SS, Swerdlow DL, Borse RH, et al. Estimating the burden of 2009 pandemic influenza A (H1N1) in the United States (April 2009–April 2010). Clin Infect Dis 2011;52(Suppl 1):S75–82.CrossRef PubMed
- Reed C, Angulo FJ, Swerdlow DL, et al. Estimates of the prevalence of pandemic (H1N1) 2009, United States, April–July 2009. Emerg Infect Dis 2009;15:2004–7. CrossRef PubMed
- Hatchett RJ, Mecher CE, Lipsitch M. Public health interventions and epidemic intensity during the 1918 influenza pandemic. Proc Natl Acad Sci U S A 2007;104:7582–7. CrossRefPubMed
- Medlock J, Meyers LA, Galvani A. Optimizing allocation for a delayed influenza vaccination campaign. PLoS Curr 2009;1:RRN1134. CrossRef PubMed
- Rainisch G, Asher J, George D, et al. Estimating Ebola treatment needs, United States [letter]. Emerg Infect Dis 2015;21:1273–5. CrossRef PubMed
- Borse RH, Shrestha SS, Fiore AE, et al. Effects of vaccine program against pandemic influenza A(H1N1) virus, United States, 2009–2010. Emerg Infect Dis 2013;19:439–48. CrossRefPubMed
- Kostova D, Reed C, Finelli L, et al. Influenza illness and hospitalizations averted by influenza vaccination in the United States, 2005–2011. PLoS One 2013;8:e66312 . CrossRef PubMed
 FIGURE. Estimated impact of delaying intervention on daily number of Ebola virus disease (Ebola) cases — Ebola Response modeling tool, Liberia, 2014–2015*
FIGURE. Estimated impact of delaying intervention on daily number of Ebola virus disease (Ebola) cases — Ebola Response modeling tool, Liberia, 2014–2015*
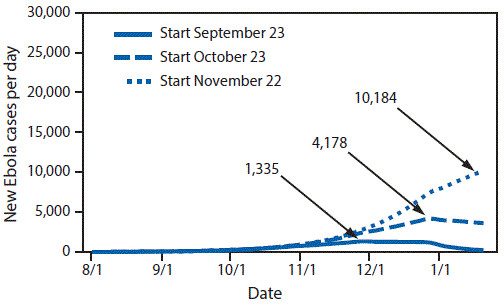
Source: Meltzer MI, Atkins CY, Knust B, et al. Estimating the future number of cases in the Ebola epidemic—Liberia and Sierra Leone, 2014–2015. MMWR Suppl 2014;63(No. Suppl 3).
* Data are not corrected for underreporting.


































No hay comentarios:
Publicar un comentario