Radiation exists all around us, from both natural and manmade sources, and is in two forms: ionizing and non-ionizing radiation.
Ionizing radiation is a form of energy that acts by removing electrons from atoms and molecules of materials that include air, water, and living tissue. Ionizing radiation can travel unseen and pass through these materials.
What is non-ionizing radiation?
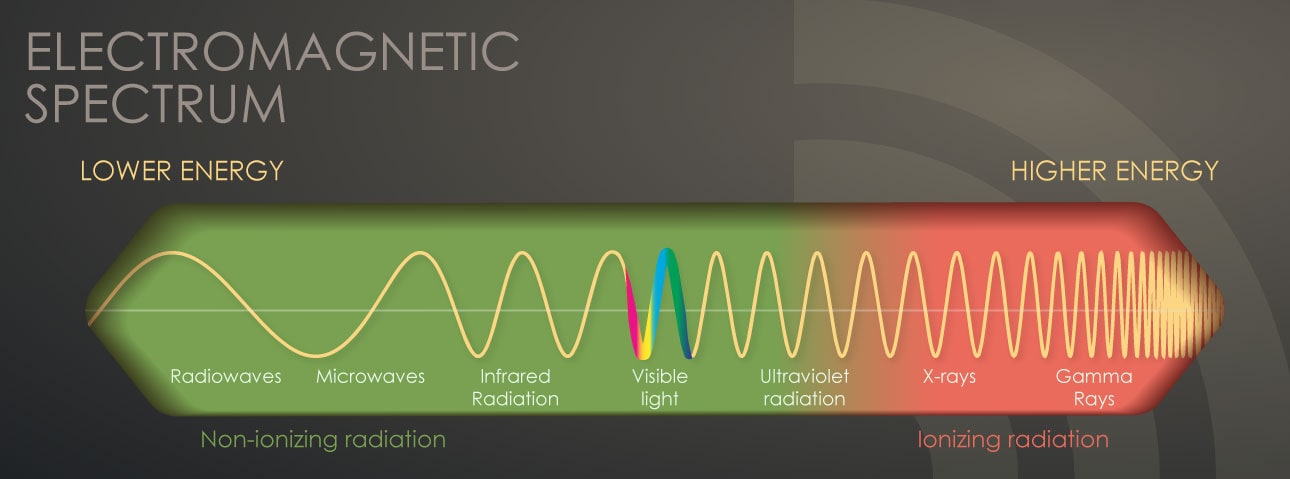
Non-ionizing radiation exists all around us from many sources. It is to the left of ionizing radiation on the electromagnetic spectrum in the figure below.
Examples include:
The dividing line between ionizing and non-ionizing radiation occurs in the ultraviolet part of the electromagnetic spectrum [shown in the illustration of the electromagnetic spectrum above]. Radiation in the ultraviolet band and at lower energies (to the left of ultraviolet) is called non-ionizing radiation, while at the higher energies to the right of the ultraviolet band is called ionizing radiation.
As we move to the left of the visible light band in the figure above, we move to lower frequencies. By “frequency” we mean how rapidly these waves move up and down. The lower the frequency, the lower the energy.
In these lower frequencies on the left side of the electromagnetic spectrum, we find infrared, microwave, radiowaves, and cell phone range radiation.
How is non-ionizing radiation different from ionizing radiation?
Put simply, non-ionizing radiation differs from ionizing radiation in the way it acts on materials like air, water, and living tissue
Unlike x-rays and other forms of ionizing radiation, non-ionizing radiation does not have enough energy to remove electrons from atoms and molecules. Non-ionizing radiation can heat substances. For example, the microwave radiation inside a microwave oven heats water and food rapidly.
What is the risk from exposure to non-ionizing radiation?
We are exposed to low levels of non-ionizing radiation every day. Exposure to intense, direct amounts of non-ionizing radiation may result in damage to tissue due to heat. This is not common and mainly of concern in the workplace for those who work on large sources of non-ionizing radiation devices and instruments.
Risk from ultraviolet (UV) radiation exposure
Ultraviolet (UV) radiation is a natural part of solar radiation, and is released by black lights, tanning beds, and electric arc lighting. Normal everyday levels of UV radiation can be helpful, and produce vitamin D. The World Health Organization (WHO) recommends 5 to 15 minutes of sun exposure 2 to 3 times a week to get enough vitamin D.
Too much UV radiation can cause skin burns, premature aging of the skin, eye damage, and skin cancer. The majority of skin cancers are caused by exposure to ultraviolet radiation.
Tanning through the use of tanning beds and tanning devices exposes the consumer to UV radiation. Exposure to tanning beds and tanning devices also increases the chance of developing skin cancer.
Risk from exposure to radiofrequency (RF) and microwave radiation
Intense, direct exposure to radiofrequency (RF) or microwave radiation may result in damage to tissue due to heat. These more significant exposures could occur from industrial devices in the workplace.


































No hay comentarios:
Publicar un comentario