
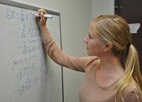 |
"FDA Invites Students to Sharpen Their Skills"
By: Nysia George, Ph.D. and Tom Powers
Biology. Chemistry. Bioinformatics. Toxicology.
Practical, hands-on laboratory work is important for all college students who want to become scientists—but, for many of them, such experiences are out of reach.
That’s one of the reasons why every summer, NCTR—FDA’s internationally acclaimed toxicological research center in Jefferson, Arkansas—hosts a special internship program for science students interested in toxicology research. Read more of the blog ...
 | Biomarkers of Tobacco Smoke Exposure |
NCTR scientists, in collaboration with colleagues from FDA's Center for Tobacco Products published an extensive review of biomarkers that has been used to monitor exposure to and damage from tobacco smoke. This review, in the current edition of Advances in Clinical Chemistry (Volume 67) examines both well-accepted biomarkers, such as urinary cotinine as well as more emerging biomarkers, such as genomic markers found in airway tissue. The review explores the range of biological matrices (urine, blood, saliva, sputum, exhalant, hair, toenails, and airway and nasal tissue) that can be used for biomarker measurements. It also contrasts biomarkers of exposure with those that measure biological dose or potential harm. The review explores the possible uses of such biomarkers, and the considerations for assay validation and biomarker qualification.
For additional information, please contact William Mattes, Ph.D., Director, Division of Systems Biology, FDA/NCTR.
Toward a Better Understanding of Vaginal Microbiota Interactions
Scientists from FDA's NCTR, Center for Food Safety and Applied Nutrition, and the Office of Regulatory Affairs' Arkansas Regional Laboratories with funding from the Office of Women’s Health have developed a defined bacterial growth medium that more closely simulates vaginal secretions for the purpose of studying bacterial interactions that occur in the vaginal tract. Lactobacillus species, which are commensal (living on or within another organism, and deriving benefit without harming or benefiting the host) with and the predominant microorganism found in a healthy vaginal tract, provide a level of protection against colonization by pathogenic bacteria using a number of mechanisms. However, this protection can be overcome byStaphylococcus aureus, leading to production of toxic shock syndrome toxin-1. This growth medium advancement allows for simultaneous growth ofLactobacillus species and ofStaphylococcus aureus. This facilitates the opportunity for study of the interaction leading to discovery of prevention strategies against menstrual cases of toxic shock syndrome. The results of this study were recently published in the Journal of Microbiological Methods.
For additional information, please contact Mark E. Hart, Ph.D., Division of Microbiology, FDA/NCTR.























.png)










No hay comentarios:
Publicar un comentario