Coronavirus Disease 2019 (COVID-19) Situation Summary
This is an emerging, rapidly evolving situation and CDC will provide updated information as it becomes available, in addition to updated guidance.
Updated February 29, 2020
Background
CDC is responding to an outbreak of respiratory disease caused by a novel (new) coronavirus that was first detected in China and which has now been detected in 60 locations internationally, including in the United States. The virus has been named “SARS-CoV-2” and the disease it causes has been named “coronavirus disease 2019” (abbreviated “COVID-19”).
On January 30, 2020, the International Health Regulations Emergency Committee of the World Health Organization declared the outbreak a “public health emergency of international concern” (PHEIC). On January 31, 2020, Health and Human Services Secretary Alex M. Azar II declared a public health emergency (PHE) for the United States to aid the nation’s healthcare community in responding to COVID-19.
Source and Spread of the Virus
Coronaviruses are a large family of viruses that are common in people and many different species of animals, including camels, cattle, cats, and bats. Rarely, animal coronaviruses can infect people and then spread between people such as with MERS-CoV, SARS-CoV, and now with this new virus (named SARS-CoV-2).
The SARS-CoV-2 virus is a betacoronavirus, like MERS-CoV and SARS-CoV. All three of these viruses have their origins in bats. The sequences from U.S. patients are similar to the one that China initially posted, suggesting a likely single, recent emergence of this virus from an animal reservoir.
Early on, many of the patients at the epicenter of the outbreak in Wuhan, Hubei Province, China had some link to a large seafood and live animal market, suggesting animal-to-person spread. Later, a growing number of patients reportedly did not have exposure to animal markets, indicating person-to-person spread. Person-to-person spread was subsequently reported outside Hubei and in countries outside China, including in the United States. Some international destinations now have apparent community spread with the virus that causes COVID-19, meaning some people have been infected who are not sure how or where they became infected. Learn what is known about the spread of newly emerged coronaviruses.
On This Page
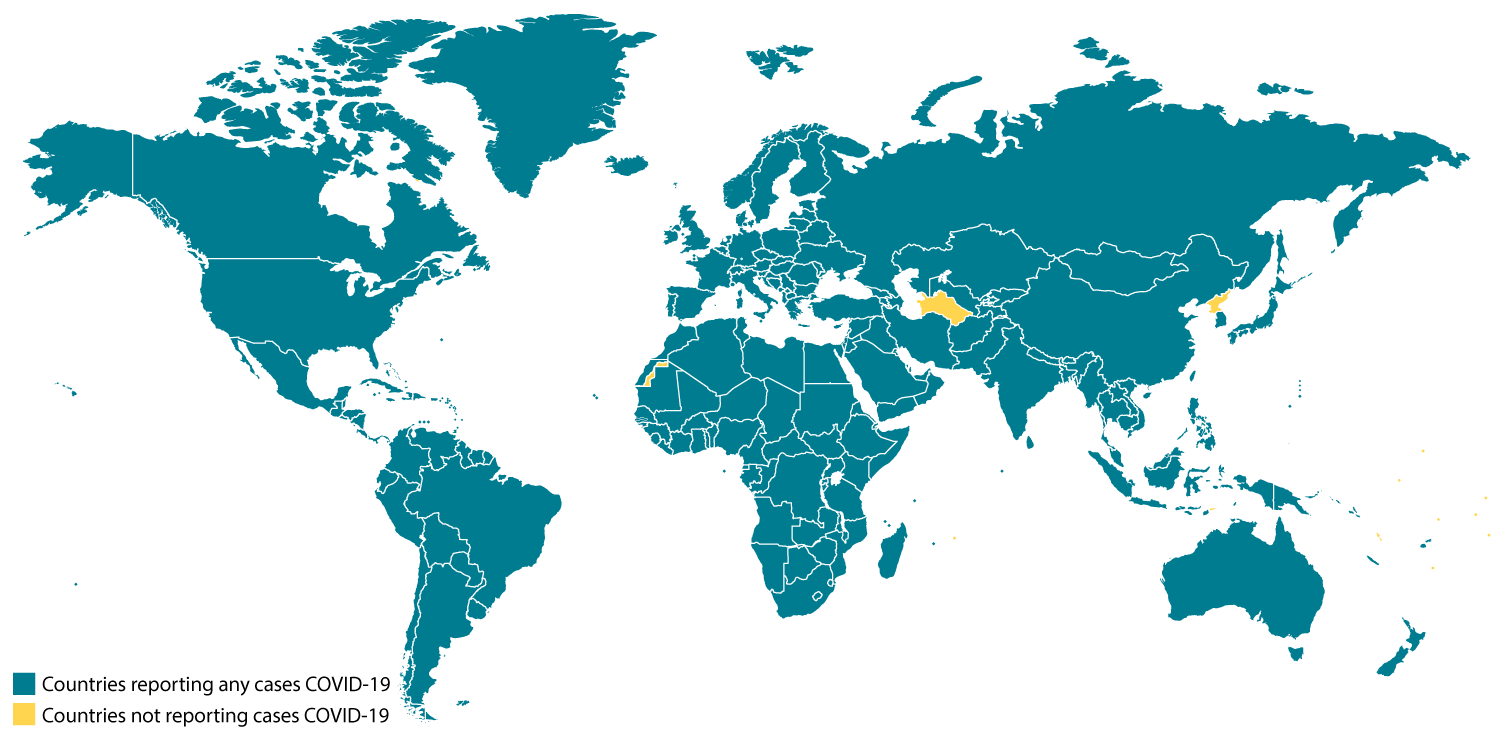
Confirmed COVID-19 Cases Global Map
Situation in U.S.
- Imported cases of COVID-19 in travelers have been detected in the U.S.
- Person-to-person spread of COVID-19 was first reported among close contacts of returned travelers from Wuhan.
- During the week of February 23, CDC reported community spread of the virus that causes COVID-19 in California (in two places), Oregon and Washington. Community spread in Washington resulted in the first death in the United States from COVID-19, as well as the first reported case of COVID-19 in a health care worker, and the first potential outbreak in a long-term care facility.
Illness Severity
Both MERS-CoV and SARS-CoV have been known to cause severe illness in people. The complete clinical picture with regard to COVID-19 is not fully understood. Reported illnesses have ranged from mild to severe, including illness resulting in death. Learn more about the symptoms associated with COVID-19.
There are ongoing investigations to learn more. This is a rapidly evolving situation and information will be updated as it becomes available.
Risk Assessment
Outbreaks of novel virus infections among people are always of public health concern. The risk from these outbreaks depends on characteristics of the virus, including how well it spreads between people, the severity of resulting illness, and the medical or other measures available to control the impact of the virus (for example, vaccine or treatment medications). The fact that this disease has caused illness, including illness resulting in death, and sustained person-to-person spread is concerning. These factors meet two of the criteria of a pandemic. As community spread is detected in more and more countries, the world moves closer toward meeting the third criteria, worldwide spread of the new virus.
While there is still much to learn about the unfolding situations in California, Oregon and Washington, preliminary information raises the level of concern about the immediate threat for COVID-19 for certain communities in the United States. The potential public health threat posed by COVID-19 is very high, to the United States and globally.
At this time, however, most people in the United States will have little immediate risk of exposure to this virus. This virus is NOT currently spreading widely in the United States. However, it is important to note that current global circumstances suggest it is likely that this virus will cause a pandemic. This is a rapidly evolving situation and the risk assessment will be updated as needed.
Current risk assessment:
- For the general American public, who are unlikely to be exposed to this virus at this time, the immediate health risk from COVID-19 is considered low.
- People in communities where ongoing community spread with the virus that causes COVID-19 has been reported are at elevated though still relatively low risk of exposure.
- Healthcare workers caring for patients with COVID-19 are at elevated risk of exposure.
- Close contacts of persons with COVID-19 also are at elevated risk of exposure.
- Travelers returning from affected international locations where community spread is occurring also are at elevated risk of exposure.
CDC has developed guidance to help in the risk assessment and management of people with potential exposures to COVID-19.
What May Happen
More cases of COVID-19 are likely to be identified in the coming days, including more cases in the United States. It’s also likely that person-to-person spread will continue to occur, including in communities in the United States. It’s likely that at some point, widespread transmission of COVID-19 in the United States will occur.
Widespread transmission of COVID-19 would translate into large numbers of people needing medical care at the same time. Schools, childcare centers, workplaces, and other places for mass gatherings may experience more absenteeism. Public health and healthcare systems may become overloaded, with elevated rates of hospitalizations and deaths. Other critical infrastructure, such as law enforcement, emergency medical services, and transportation industry may also be affected. Health care providers and hospitals may be overwhelmed. At this time, there is no vaccine to protect against COVID-19 and no medications approved to treat it. Nonpharmaceutical interventions would be the most important response strategy.
CDC Response
Global efforts at this time are focused concurrently on containing the spread and mitigating the impact of this virus. The federal government is working closely with state, local, tribal, and territorial partners, as well as public health partners, to respond to this public health threat. The public health response is multi-layered, with the goal of detecting and minimizing introductions of this virus in the United States. CDC is operationalizing all of its pandemic preparedness and response plans, working on multiple fronts to meet these goals, including specific measures to prepare communities to respond to local transmission of the virus that causes COVID-19. There is an abundance of pandemic guidance developed in anticipation of an influenza pandemic that is being repurposed and adapted for a COVID-19 pandemic.
Highlights of CDC’s Response
- CDC established a COVID-19 Incident Management System on January 7, 2020. On January 21, CDC activated its Emergency Operations Center to better provide ongoing support to the COVID-19 response.
- The U.S. government has taken unprecedented steps with respect to travel in response to the growing public health threat posed by this new coronavirus:
- Effective February 2, at 5pm, the U.S. government suspended entry of foreign nationals who have been in China within the past 14 days.
- U.S. citizens, residents, and their immediate family members who have been in Hubei province and other parts of mainland China are allowed to enter the United States, but they are subject to health monitoring and possible quarantine for up to 14 days.
- Effective February 2, at 5pm, the U.S. government suspended entry of foreign nationals who have been in China within the past 14 days.
- On February 29, the U.S. government announced it was suspending entry of foreign nationals who have been in Iran within the past 14 days.
- CDC has issued the following travel guidance related to COVID-19:
- China — Level 3, Avoid Nonessential Travel — updated February 22;
- Hong Kong — Level 1, Practice Usual Precautions — issued February 19;
- Iran — Level 3, Avoid Nonessential Travel — updated February 28;
- Italy — Level 3, Avoid Nonessential Travel — updated February 28;
- Japan — Level 2, Practice Enhanced Precautions — updated February 22;
- South Korea — Level 3, Avoid Nonessential Travel — updated February 24.
- CDC also recommends that all travelers reconsider cruise ship voyages into or within Asia at this time.
- CDC has issued the following travel guidance related to COVID-19:
- CDC is issuing clinical guidance, including:
- On January 30, CDC published guidance for healthcare professionals on the clinical care of COVID-19 patients.
- On February 3, CDC posted guidance for assessing the potential risk for various exposures to COVID-19 and managing those people appropriately.
- On February 27, CDC updated its criteria to guide evaluation of persons under investigation for COVID-19.
- On February 28, CDC issued a Health Alert Network (HAN): Update and Interim Guidance on Outbreak of COVID-19.
- CDC has deployed multidisciplinary teams to support state health departments case identification, contact tracing, clinical management, and communications.
- CDC has worked with the Department of State, supporting the safe return of Americans who have been stranded as a result of the ongoing outbreaks of COVID-19 and related travel restrictions. CDC has worked to assess the health of passengers as they return to the United States and provided continued daily monitoring of people who are quarantined.

This is a picture of CDC’s laboratory test kit for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). CDC is shipping the test kits to laboratories CDC has designated as qualified, including U.S. state and local public health laboratories, Department of Defense (DOD) laboratories and select international laboratories. The test kits are bolstering global laboratory capacity for detecting SARS-CoV-2.
- An important part of CDC’s role during a public health emergency is to develop a test for the pathogen and equip state and local public health labs with testing capacity.
- After distribution of a CDC rRT-PCR test to diagnose COVID-19 to state and local public health labs started, performance issues were identified related to a problem in the manufacturing of one of the reagents. Laboratories were not able to verify the test performance.
- CDC worked on two potential resolutions to this problem.
- CDC developed a new protocol that uses two of the three components of the original CDC test kit to detect the virus that causes COVID-19 after establishing that the third component, which was the problem with the original test, can be excluded from testing without affecting accuracy. CDC is working with FDA to amend the existing Emergency Use Authorization (EUA) for the test, but in the meantime, FDA granted discretionary authority for the use of the original test kits.
- Public health laboratories can use the original CDC test kit to test for the virus that causes COVID-19 using the new protocol.
- Further, newly manufactured kits have been provided to the International Reagent Resource for distribution.
- CDC developed a new protocol that uses two of the three components of the original CDC test kit to detect the virus that causes COVID-19 after establishing that the third component, which was the problem with the original test, can be excluded from testing without affecting accuracy. CDC is working with FDA to amend the existing Emergency Use Authorization (EUA) for the test, but in the meantime, FDA granted discretionary authority for the use of the original test kits.
- Combined with other reagents that CDC has procured, there are enough testing kits to test more than 75,000 people.
- In addition, CDC has two laboratories conducting testing for the virus that causes COVID-19. CDC can test approximately 350 specimens per day.
- Commercial labs are working to develop their own tests that hopefully will be available soon. This will allow a greater number of tests to happen close to where potential cases are.
- CDC has grown the COVID-19 virus in cell culture, which is necessary for further studies, including for additional genetic characterization. The cell-grown virus was sent to NIH’s BEI Resources Repositoryexternal icon for use by the broad scientific community.
CDC Recommends
- Everyone can do their part to help us respond to this emerging public health threat:
- It’s currently flu and respiratory disease season and CDC recommends getting a flu vaccine, taking everyday preventive actions to help stop the spread of germs, and taking flu antivirals if prescribed.
- If you are a healthcare provider, be on the look-out for:
- People who recently traveled from China or another affected area and who have symptoms associated with COVID-19, and
- People who have been in close contact with someone with COVID-19 or pneumonia of unknown cause. (Consult the most recent definition for patients under investigation [PUIs].)
- If you are a healthcare provider or a public health responder caring for a COVID-19 patient, please take care of yourself and follow recommended infection control procedures.
- If you are a close contact of someone with COVID-19 and develop symptoms of COVID-19, call your healthcare provider and tell them about your symptoms and your exposure.
- If you are a resident in a community where person-to-person spread of COVID-19 has been detected and you develop COVID-19 symptoms, call your healthcare provider and tell them about your symptoms.
- For people who are ill with COVID-19, but are not sick enough to be hospitalized, please follow CDC guidance on how to reduce the risk of spreading your illness to others. People who are mildly ill with COVID-19 are able to isolate at home during their illness.
- If you have been in China or another affected area or have been exposed to someone sick with COVID-19 in the last 14 days, you will face some limitations on your movement and activity for up to 14 days. Please follow instructions during this time. Your cooperation is integral to the ongoing public health response to try to slow spread of this virus.
Other Available Resources
The following resources are available with information on COVID-19























.png)












No hay comentarios:
Publicar un comentario