


Drug Safety Communication: Updated FDA review concludes that use of type 2 diabetes medicine pioglitazone may be linked to an increased risk of bladder cancerAs a result of an updated review, FDA has concluded that use of the type 2 diabetes medicine pioglitazone (Actos, Actoplus Met, Actoplus Met XR, Duetact, Oseni) may be linked to an increased risk of bladder cancer. The labels of pioglitazone-containing medicines already contain warnings about this risk, and we have now approved label updates to describe the additional studies we reviewed. We alerted the public about the possible risk of bladder cancer in September 2010 and June 2011 based on interim results from a 10-year epidemiologic study. We changed the labels of pioglitazone-containing medicines in August 2011 to include warnings about this risk, and required the manufacturer to modify and continue the 10-year study. More information
Drug Safety Communication: General Anesthetic and Sedation Drugs - New Warnings for Young Children and Pregnant WomenFDA is warning that repeated or lengthy use of general anesthetic and sedation drugs during surgeries or procedures in children younger than 3 years or in pregnant women during their third trimester may affect the development of children’s brains. Consistent with animal studies, recent human studies suggest that a single, relatively short exposure to general anesthetic and sedation drugs in infants or toddlers is unlikely to have negative effects on behavior or learning. However, further research is needed to fully characterize how early life anesthetic exposure affects children’s brain development. To better inform the public about this potential risk, we are requiring warnings to be added to the labels of general anesthetic and sedation drugs (see List of General Anesthetic and Sedation Drugs Affected by this Label Change). We will continue to monitor the use of these drugs in children and pregnant women and will update the public if additional information becomes available. More information
Drug Safety Communication: Chantix (varenicline) and Zyban (bupropion) - Mental Health Side Effects RevisedBased on an FDA review of a large clinical trial that FDA required the drug companies to conduct, FDA determined the risk of serious side effects on mood, behavior, or thinking with the stop-smoking medicines Chantix (varenicline) and Zyban (bupropion) is lower than previously suspected. The risk of these mental health side effects is still present, especially in those currently being treated for mental illnesses such as depression, anxiety disorders, or schizophrenia, or who have been treated for mental illnesses in the past. However, most people who had these side effects did not have serious consequences such as hospitalization. The results of the trial confirm that the benefits of stopping smoking outweigh the risks of these medicines. See the Drug Safety Communication for a data summary. More information
FDA expands indication for continuous glucose monitoring system, first to replace fingerstick testing for diabetes treatment decisions
FDA has expanded the approved use of Dexcom’s G5 Mobile Continuous Glucose Monitoring System to allow for replacement of fingerstick blood glucose (sugar) testing for diabetes treatment decisions in people 2 years of age and older with diabetes. This is the first FDA-approved continuous glucose monitoring system that can be used to make diabetes treatment decisions without confirmation with a traditional fingerstick test. The system was previously approved to complement, not replace, fingerstick testing for diabetes treatment decisions. More information |
Comunicaciones de la FDA sobre la seguridad de los medicamentos en español
Descargo de responsabilidad: La FDA reconoce la necesidad de proporcionar información importante sobre seguridad de los medicamentos en idiomas distintos al inglés. Hacemos lo mejor posible para proporcionar versiones en español precisas y oportunas de nuestras Comunicaciones de Seguridad de Medicamentos. Sin embargo, en caso que existiera discrepancias entre las versiones en inglés y la de español, la información contenida en la versión en inglés es la que se considera como versión oficial. Si tiene alguna pregunta, por favor contáctese con Division of Drug Information en druginfo@fda.hhs.gov. Comunicaciones de la FDA |


FDA knows the major public health consequences that can result from drug shortages. These shortages occur for many reasons including manufacturing and quality problems, delays and discontinuations. When issues are discovered, FDA works closely with the company to address risks involved to prevent harm to patients. FDA also considers the impact a shortage would have on patient care and access and works with the company to restore supplies while also ensuring safety for patients. More information
|
Drug Shortages Voluntarily Reported by Manufacturers During the Past 2 Weeks:
Information about blood and biologic shortages, resolved shortages, and discontinuations
La FDA reconoce las consecuencias significativas para la salud pública que pueden resultar de la escasez de medicamentos y hace un gran esfuerzo dentro de sus facultades legales para abordar yprevenir la escasez de medicamentos. La escasez se produce por muchas razones, incluyendo problemas de fabricación y calidad, retrasos y discontinuación del producto. Cuando los problemas son descubiertos por la empresa o el público y reportados a la FDA o se descubren por inspecciones de la FDA, la FDA trabaja en estrecha colaboración con la empresa para hacer frente a los riesgosinvolucrados y evitar daños a los pacientes. La FDA también considera el impacto que una escaseztendría en la atención médica del paciente y al acceso del producto y trabaja con la empresa pararestablecer el suministro al tiempo que garantiza la seguridad de los pacientes. Más información
|
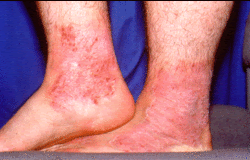
FDA approves Eucrisa for eczema
FDA has approved Eucrisa (crisaborole) ointment to treat mild to moderate eczema (atopic dermatitis) in patients two years of age and older. Atopic dermatitis, a chronic inflammatory skin disease, is often referred to as "eczema," which is a general term for the several types of inflammation of the skin. Atopic dermatitis is the most common of the many types of eczema and onset typically begins in childhood and can last through adulthood. The cause of atopic dermatitis is a combination of genetic, immune and environmental factors. In atopic dermatitis, the skin develops red, scaly and crusted bumps, which are extremely itchy. Scratching leads to swelling, cracking, "weeping" clear fluid, and finally, coarsening and thickening of the skin. More information
FDA grants accelerated approval to new treatment for advanced ovarian cancer
FDA has granted accelerated approval to Rubraca (rucaparib) to treat women with a certain type of ovarian cancer. Rubraca is approved for women with advanced ovarian cancer who have been treated with two or more chemotherapies and whose tumors have a specific gene mutation (deleterious BRCA) as identified by an FDA-approved companion diagnostic test. The agency approved Rubraca under its accelerated approval program, which allows approval of a drug to treat a serious or life-threatening disease or condition based on clinical data showing the drug has an effect on a surrogate (substitute) endpoint that is reasonably likely to predict clinical benefit. The sponsor is continuing to study this drug in patients with advanced ovarian cancer who have BRCA gene mutations and in patients with other types of ovarian cancer. The FDA also granted the Rubraca application breakthrough therapy designation and priority review status. Rubraca also received orphan drug designation, which provides incentives such as tax credits, user fee waivers and eligibility for exclusivity to assist and encourage the development of drugs intended to treat rare diseases. More information
FDA approves first autologous cellularized scaffold for the repair of cartilage defects of the knee
FDA has approved Maci (autologous cultured chondrocytes on porcine collagen membrane) for the repair of symptomatic, full-thickness cartilage defects of the knee in adult patients. Maci is the first FDA-approved product that applies the process of tissue engineering to grow cells on scaffolds using healthy cartilage tissue from the patient’s own knee. Knee problems are common, and occur in people of all ages. Cartilage defects in the knee can result from an injury, straining the knee beyond its normal motion, or can be caused by overuse, muscle weakness, and general wear and tear. More information |
For information on drug approvals or to view prescribing information and patient information, please visit Drugs@FDA or DailyMed.

View FDA's Comments on Current Draft Guidance page, for a list of current draft guidances and other topics of interest for patients and caregivers.
|

FDA’s Naloxone App Prize Competition Celebrates Innovation In Search of Technological Solutions to the Opioid Epidemic, by Peter Lurie, M.D., M.P.H., Associate Commissioner for Public Health Strategy and Analysis
The epidemic of opioid dependence and abuse has had an impact upon communities both large and small across the United States. Since 1999, rates of overdose deaths involving opioids, whether prescription painkillers or street drugs like heroin, have nearly quadrupled. In 2015, 91 Americans died from an opioid overdose each day. Many local, state, and federal agencies have worked to prevent these tragic deaths by expanding access to naloxone, a prescription medication that can rapidly reverse the effects of an opioid overdose. Supporting increased access to naloxone is also a key objective of the multifaceted Opioid Action Plan released by FDA earlier this year. In addition, FDA has sponsored two national meetings on the topic and in recent years has approved two naloxone products that facilitate use by laypeople.
To read the rest of this post, see FDA Voice Blog, December 14, 2016.
|

21st Century Cures Act: Making Progress on Shared Goals for Patients, by Robert M. Califf, M.D., Commissioner FDA
President Obama has signed into law the 21st Century Cures Act, which, I am pleased to report, builds on FDA’s ongoing efforts to advance medical product innovation and ensure that patients get access to treatments as quickly as possible, with continued assurance from high quality evidence that they are safe and effective. Cures will greatly improve FDA’s ability to hire and retain scientific experts. One of our ongoing challenges has been recruiting and retaining the experts we need in specialized areas to allow us to get our work done and meet our growing responsibilities. This is an especially important need given the tremendous advances in biological sciences, engineering, information technology and data science. Preventive, diagnostic and therapeutic strategies will become more complex with much greater potential for benefit and in some cases greater risk if used without adequate evidence to exclude risks that exceed potential benefits.
To read the rest of this post, see FDA Voice Blog, December 13, 2016.
|

Faster Information from FDA Means Improved Drug Safety for Patients, by Mary E. Kremzner, PharmD, MPH, CAPT, U.S. Public Health Service, Director, Division of Drug Information, in FDA’s Center for Drug Evaluation and Research
FDA is now making it easier and faster for health care professionals and patients to get the most up-to-date drug safety information on the more than 18,000 drugs available on our website. Our improved Drug Safety Labeling Changes Program enables FDA to post the latest safety information about a medicine almost at the same time we approve a change, as opposed to once a month. When your physician or other health care professional prescribes your medicine using their e-prescribing system, the new safety information now displays much faster than with our previous system. Here’s how – within days of FDA approval of new drug safety information for a drug product, the information is entered into the safety labeling changes database. Health IT vendors that provide clinical and drug information support for hospitals and pharmacies are then alerted to integrate the safety labeling changes data into their systems. So instead of waiting weeks for the monthly release of all safety labeling updates, this information is now accessible in days.
To read the rest of this post, see FDA Voice Blog, December 12, 2016.
|
 |
Director’s Corner Podcasts
The Director’s Corner is an audio podcast series featuring the director of FDA’s Center for Drug Evaluation and Research and produced by CDER’s Office of Communications. In the latest podcast, Dr. Janet Woodcock discusses the FDA's actions to implement the Opioid Action Plan, including important safety labeling changes. Listen to this podcast Addressing the Opioid Epidemic or read the Transcript: Addressing the Opioid Epidemic. |

In this section you will find a comprehensive list of all the meetings that the FDA is involved with. The meetings may include advisory committee meetings, public workshops and public conferences that are seeking to hear from patients and caregivers.
Most FDA meetings are free to the public and do not require the public to register. Interested persons may present data, information, or views, orally at the meeting, or in writing, on issues pending before the committee. Other types of meetings listed may require prior registration and fees.
|
View FDA's Patient Network Calendar of Public Meetings page for a complete list of meetings and workshops.
For additional information on other agency meetings please visit Meetings, Conferences, & Workshops.
|

LASIK Quality of Life Collaboration Project
In October 2009, the FDA, the National Eye Institute (NEI), and the Department of Defense (DoD) launched the LASIK Quality of Life Collaboration Project (LQOLCP) to help better understand the potential risk of severe problems that can result from LASIK. The project aimed to develop a tool to determine the percent of patients who develop difficulties performing their usual activities following LASIK, and to identify predictors for those patients. At the time we developed our project, there was a limited amount of valid scientific data on certain patient-reported outcomes (PROs) related to LASIK. A PRO is a report of a condition experienced and reported by the patient, not the health care provider. Most LASIK studies used tools, such as questionnaires, to assess visual symptoms, but only after the surgery. The Patient-Reported Outcomes with LASIK (PROWL) studies in the LQOLCP assessed visual symptoms both before and after their LASIK surgery to identify changes over time. The studies also measured the impact symptoms directly had on performing usual activities, which had not previously been done. More information |
How to Report Product Problems and Complaints to the FDA
FDA protects the public health by helping to ensure the safety, efficacy, and security of human and veterinary drugs, biological products, medical devices, and products that emit radiation—and by helping to ensure the safety and security of our nation’s food supply and cosmetic products. The FDA also regulates tobacco products. If you have a problem or concern about any of the products that the agency regulates, the FDA wants to hear from you. More information
FDA protects the public health by helping to ensure the safety, efficacy, and security of human and veterinary drugs, biological products, medical devices, and products that emit radiation—and by helping to ensure the safety and security of our nation’s food supply and cosmetic products. The FDA also regulates tobacco products. If you have a problem or concern about any of the products that the agency regulates, the FDA wants to hear from you. More information
More Consumer Updates
For previously published Consumer Update articles that are timely and easy-to-read and cover all FDA activities and regulated products. More information
For previously published Consumer Update articles that are timely and easy-to-read and cover all FDA activities and regulated products. More information
En Español
La información en esta página es para el público en general, y para profesionales y educadores de salud. Esta información puede ser distribuida y publicada sin previa autorización. En Español
La información en esta página es para el público en general, y para profesionales y educadores de salud. Esta información puede ser distribuida y publicada sin previa autorización. En Español

How to Cut Food Waste and Maintain Food Safety
Food safety is a major concern. The Centers for Disease Control and Prevention (CDC) estimates that there are about 48 million cases of foodborne illness annually – the equivalent of sickening 1 in 6 Americans each year. And each year these illnesses result in an estimated 128,000 hospitalizations and 3,000 deaths. Food waste is also a major concern. Wasted food is a huge challenge to our natural resources, our environment, and our pocketbooks. More information
The Safety Reporting Portal
The Safety Reporting Portal (SRP) streamlines the process of reporting product issues to the Food and Drug Administration and the National Institutes of Health. Whatever your role, (manufacturer, health care professional, researcher, public health official, or concerned citizen), when you submit a safety report through this Portal, you make a vital contribution to the safety of America's food supply, medicines, and other products that touch us all. More information
Center for Food Safety and Applied NutritionThe Center for Food Safety and Applied Nutrition, known as CFSAN, carries out the mission of FDA. The Center provides services to consumers, domestic and foreign industry and other outside groups regarding field programs; agency administrative tasks; scientific analysis and support; and policy, planning and handling of critical issues related to food and cosmetics. More information
Food Facts for YouThe Center for Food Safety and Applied Nutrition, known as CFSAN, issues food facts for consumers to keep you and your family safe. More information
|

Keep Your Dogs and Cats Safe From Holiday Hazards
This holiday season, while you’re busy decorating, cooking, and wrapping gifts, remember to watch out for holiday temptations for your pets. FDA veterinarian Carmela Stamper tells how to keep your animals safe. If your dog received a stocking full of pet treats, make sure he doesn’t gobble them all up at once. According to Stamper, if he eats the treats whole, or eats too many at once, he may not be able to digest them. Unchewed pet treats can get stuck in the trachea (windpipe) or gastrointestinal tract (esophagus, stomach, and intestines), particularly in small dogs. If your dog is in obvious distress from eating too much too fast, says Stamper, contact your vet immediately. Some telltale signs are drooling, choking, or vomiting. More information
Animal Health Literacy
Animal Health Literacy means timely information for the benefit of all animals and their humans. With continuous communication and outreach, the Center for Veterinary Medicine (CVM) strives to enhance the public trust, promote safe and effective use of the animal health products we regulate, and share our scientific endeavors. CVM provides reliable, science-based information to promote animal and human health. More information and Publicaciones en Español del FDA
Animal and Veterinary Updates
Animal and veterinary updates provide information to keep your pets healthy and safe. More information |

How to Report a Pet Food Complaint
You can report complaints about a pet food product electronically through the Safety Reporting Portal or you can call your state’s FDA Consumer Complaint Coordinators. Please provide as much information as possible in your complaint, such as exact name of product, type of container, lot number, UPC codes, how the food was stored, and purchase date and exact location where purchased. If possible, please save the original packaging until the pet food has been consumed. The packaging contains IMPORTANT information often needed to identify the variety of pet food, the manufacturing plant, and the production date. More information |
FDA takes action against four tobacco manufacturers for illegal sales of flavored cigarettes labeled as little cigars or cigars
FDA has issued warning letters to four tobacco manufacturers — Swisher International Inc., Cheyenne International LLC, Prime Time International Co. and Southern Cross Tobacco Company Inc. — for selling flavored cigarettes that are labeled as little cigars or cigars, which is a violation of the Family Smoking Prevention and Tobacco Control Act. The companies received warning letters for products under the “Swisher Sweets,” Cheyenne,” “Prime Time” and “Criss-Cross” brands in a variety of youth-appealing flavors, including grape, cherry, wild cherry and strawberry. “Flavored cigarettes appeal to kids and disguise the bad taste of tobacco, but they are just as addictive as regular tobacco products and have the same harmful health effects,” said Mitch Zeller, J.D., director of the FDA’s Center for Tobacco Products. “Because about 90 percent of adult daily smokers smoked their first cigarette by the age of 18, continued enforcement of the ban on cigarettes with characterizing flavors is vital to protect future generations from a lifetime of addiction.” More information
FDA takes action on applications seeking to market modified risk tobacco products
FDA has taken action on the first applications reviewed through the modified risk tobacco product (MRTP) pathway for eight Swedish Match North America Inc. snus smokeless tobacco products sold under the General brand name. The agency denied the company’s request to remove a currently required warning stating that the products can cause gum disease and tooth loss. With respect to the company’s other requests to remove or revise two additional currently required warnings, the agency deferred final action and issued a response that offers the company an option to amend its applications.While all tobacco products pose risks, the MRTP pathway outlined in the 2009 Family Smoking Prevention and Tobacco Control Act allows companies to submit applications for the FDA to evaluate whether a tobacco product may be sold or distributed for use to reduce harm or the risk of tobacco-related disease associated with commercially marketed tobacco products. More information |
FDA’s LGBT Public Education Campaign Makes Headlines
FDA’s “This Free Life” campaign, designed to prevent and reduce tobacco use among LGBT young adults ages 18-24, recently won the Multicultural Award of Excellence in the LGBT category at the annual Association of National Advertisers (ANA) Multicultural Marketing & Diversity Conference. Watch the powerful ad and share it with someone you know in the LGBT community to encourage them to live tobacco free!
FDA’s “This Free Life” campaign, designed to prevent and reduce tobacco use among LGBT young adults ages 18-24, recently won the Multicultural Award of Excellence in the LGBT category at the annual Association of National Advertisers (ANA) Multicultural Marketing & Diversity Conference. Watch the powerful ad and share it with someone you know in the LGBT community to encourage them to live tobacco free!
How Cigarettes are Made and How You Can Make a Plan to QuitEver wonder why it’s so hard to kick smoking to the curb? Well, we’ve got an answer! CTP recently introduced an infographic that explores how cigarettes are made, highlighting how their very design may be an obstacle to quitting. As people across the country pledge to quit smoking, consider sharing this infographic with someone who could benefit.

Center for Tobacco Products updated safety reporting portal
CTP recently updated its Safety Reporting Portal, an online tool for reporting suspected problems with tobacco products. Whether you’re a consumer, manufacturer, clinical investigator, or health professional, let FDA know about products that seem to be damaged, defective, contaminated, or that smell or taste wrong. More information and to report a safety concern online.
Help FDA Protect Americans from Unlawful Tobacco Product ViolationsHave you witnessed a potential tobacco product violation, such as a sale to someone under 18 years of age, flavored cigarette sales, or vending machines that give minors access to tobacco products? We hope you will report these and other potential violations to FDA.
|
 |
Missed the Last Issue of CTPConnect?
FDA’s Center for Tobacco Products (CTP) publishes a regular periodical known as CTPConnect, a plainspoken digest with the latest stories from the Center. Sign up today to receive the next issue of CTPConnect and other important updates from CTP directly to your inbox. |

Bad reaction to a cosmetic? FDA needs to know
What do you do if you have a reaction after using a cosmetic product? First, stop using the product and contact your healthcare provider. Next, please report it to FDA. Here’s why that next step is so important: Cosmetic products aren’t required by law to have FDA approval before they go on the market. Companies that market cosmetics have a legal responsibility to ensure product safety, but FDA can only take action if the product is shown to be unsafe after the product is on the market. And, because the law doesn’t require cosmetic companies to share customer complaints or other safety information with FDA, voluntary reports from consumers and healthcare providers are one of the best ways for FDA to learn about any problems. Cosmetics include a range of products people use every day, such as moisturizers, makeup, shampoos and conditioners, face and body washes, deodorants, nail care products, hair dyes and relaxers, and tattoos. More information |
Recalls and Alerts
To see safety alerts and recent recalls related to cosmetics and other products regulated by FDA. More information
To see safety alerts and recent recalls related to cosmetics and other products regulated by FDA. More information
What to watch for, how to report
You can report an allergic reaction, a rash, redness, burn, hair loss, headache, infection, illness, or any other unexpected reaction, whether or not it required medical treatment. You can also report a bad smell, color change, or other sign of contamination. You can choose the way you’d prefer to report:
You can report an allergic reaction, a rash, redness, burn, hair loss, headache, infection, illness, or any other unexpected reaction, whether or not it required medical treatment. You can also report a bad smell, color change, or other sign of contamination. You can choose the way you’d prefer to report:
- Call an FDA Consumer Complaint Coordinator if you want to speak directly to a person about your problem.
- Complete an electronic Voluntary MedWatch form online.
- Complete a paper Voluntary MedWatch form that you can mail to FDA.

Information about Expanded Access
Expanded access, sometimes called "compassionate use," is the use outside of a clinical trial of an investigational medical product (i.e., one that has not been approved by FDA). FDA is committed to increasing awareness of and knowledge about its expanded access programs and the procedures for obtaining access to human investigational drugs (including biologics) and medical devices. More information |
Learn about what your physician should do before submitting a request for individual patient expanded access use of an investigational medical product, who may be eligible for expanded access, associated costs, FDA contacts and more. Information for Patients
|
Learn about your responsibilities under the expanded access pathway, how to submit a request for expanded access for an individual patient (including for emergency use), which forms to use, FDA contacts and more. Information for Physicians
|

FDA Patient Network
The FDA Patient Network contains a series of webpages, webinars and presentations on topics related to patient engagement, FDA regulations, understanding medical product (Drugs, Biologics, and Devices) approval and medical product safety updates, Take me to the FDA Patient Network or take me to FDA Webinars.
FDA Basics
Each month, different centers and offices at FDA will host an online session where the public can ask questions to senior FDA officials about a specific topic or just listen in to learn more about FDA. More information
Educational Videos
|

healthfinder.gov
Welcome to healthfinder.gov, a government Web site where you will find information and tools to help you and those you care about stay healthy. More information /más información
FDA Email Updates
Sign up for one of the FDA disease specific e-mail list that delivers updates, including product approvals, safety warnings, notices of upcoming meetings, and notices on proposed regulatory guidances. |





















.png)












No hay comentarios:
Publicar un comentario