A new DRUG TRIALS SNAPSHOT is now available.

Drug Trials Snapshots: SARCLISA
SARCLISA is a drug used to treat a form of blood cancer called multiple myeloma. SARCLISA is for adult patients whose cancer returned or did not respond to, at least two previous treatments for multiple myeloma.
It is to be used in combination with two other drugs: Dexamethasone (a type of corticosteroid) and pomalidomide.
SARCLISA is an injection. It is given by a healthcare profesional directly into the vein (intravenous infusion) every week for 4 weeks followed by every 2 weeks.
See more Drug Trials Snapshots or contact us with questions at Snapshots@fda.hhs.gov.
SARCLISA (isatuximab-irfc)
(sar-cli-sa)
Sanofi-Aventis U.S. LLC
Approval date: March 2, 2020
(sar-cli-sa)
Sanofi-Aventis U.S. LLC
Approval date: March 2, 2020
DRUG TRIALS SNAPSHOT SUMMARY:
What is the drug for?
SARCLISA is a drug used to treat a form of blood cancer called multiple myeloma. SARCLISA is for adult patients whose cancer returned or did not respond to, at least two previous treatments for multiple myeloma.
It is to be used in combination with two other drugs:
- dexamethasone (a type of corticosteroid) and
- pomalidomide.
How is this drug used?
SARCLISA is an injection. It is given by a healthcare profesional directly into the vein (intravenous infusion) every week for 4 weeks followed by every 2 weeks.
What are the benefits of this drug?
In the trial, patients who received SARCLISA (in combination with pomalidomadie and dexamethasone) lived longer without the cancer growing (average about 12 months) in comparison to patients who received pomalidomadie and dexamethasone combination only (average about 6 months).
Were there any differences in how well the drug worked in clinical trials among sex, race and age?
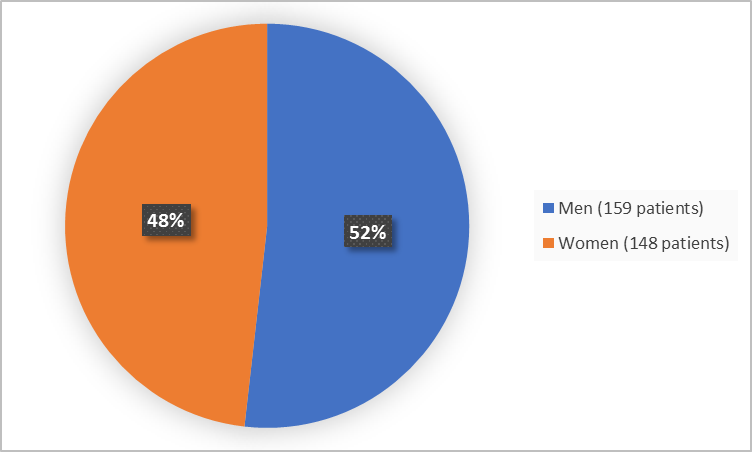
- Sex: SARCLISA worked similarly in men and women.
- Race: The majority of patients were White. Differences in response to SARCLISA among different races could not be determined.
- Age: SARCLISA worked similarly in patients below and above 65 years of age.
What are the possible side effects?
SARCLISA may cause serious side effects including infusion reactions, low neutrophil count (neutropenia), and possible development of new cancers. The most common side effects of SARCLISA are low blood counts, infusion reactions, pneumonia, upper respiratory infections and diarrhea.
Were there any differences in side effects among sex, race and age?
- Sex: The occurrence of overall side effects was similar in men and women. The occurrence of infections was higher in men than women.
- Race: The majority of patients were White. Differences in side effects among races could not be determined.
- Age: The occurrence of overall side effect was similar in patients younger and older than 65 years. The occurence of infections was higher in patients older than 65 years of age.
WHO WAS IN THE CLINICAL TRIALS?
Who participated in the clinical trials?
The FDA approved SARCLISA based on evidence from a clinical trial (NCT02990338) of 307 patients with previously treated multiple myeloma. The trial was conducted at 102 sites in Europe, North America, Asia, Australia and New Zealand.
Figure 1 summarizes how many men and women were in the clinical trial.
Figure 1. Baseline Demographics by Sex (efficacy population)
FDA Review
Figure 2 and Table 1 summarize the percentage of patients by race in the clinical trial.
Figure 2. Baseline Demographics by Race (efficacy population)
*Includes Native Hawaiian or other Pacific Islander,missing and not reported FDA Review
Figure 3 summarizes the percentage of patients by age group in the clinical trial.
Figure 3. Baseline Demographics by Age (efficacy population)
FDA Review
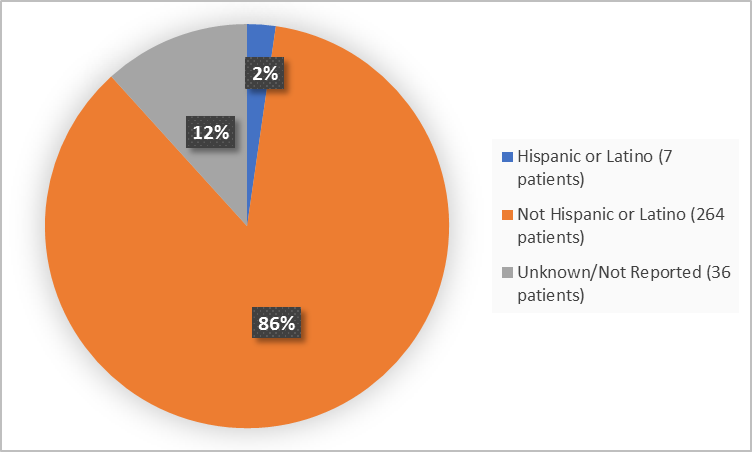
Figure 4 summarizes the percentage of patients by ethnicity in the clinical trial.
Figure 4. Baseline Demographics by Ethnicity (efficacy population)
FDA Review
How were the trials designed?
There was one trial that evaluated the efficacy and side effects of SARCLISA in patients with previously treated multiple myeloma. Patients were randomly assigned to receive either SARCLISA (in combination with pomalidomide and low-dose dexamethasone) or active comparator (pomalidomide and low-dose dexamethasone).Treatment was administered in both groups in 28-day cycles until disease progression or unacceptable toxicity. Both patients and health care providers knew which treatment was given.
The trial measured the time patients lived without the cancer growing (progression-free survival or PFS).
GLOSSARY
CLINICAL TRIAL: Voluntary research studies conducted in people and designed to answer specific questions about the safety or effectiveness of drugs, vaccines, other therapies, or new ways of using existing treatments.
COMPARATOR: A previously available treatment or placebo used in clinical trials that is compared to the actual drug being tested.
EFFICACY: How well the drug achieves the desired response when it is taken as described in a controlled clinical setting, such as during a clinical trial.
PLACEBO: An inactive substance or “sugar pill” that looks the same as, and is given the same way as, an active drug or treatment being tested. The effects of the active drug or treatment are compared to the effects of the placebo.
SUBGROUP: A subset of the population studied in a clinical trial. Demographic subsets include sex, race, and age groups.
COMPARATOR: A previously available treatment or placebo used in clinical trials that is compared to the actual drug being tested.
EFFICACY: How well the drug achieves the desired response when it is taken as described in a controlled clinical setting, such as during a clinical trial.
PLACEBO: An inactive substance or “sugar pill” that looks the same as, and is given the same way as, an active drug or treatment being tested. The effects of the active drug or treatment are compared to the effects of the placebo.
SUBGROUP: A subset of the population studied in a clinical trial. Demographic subsets include sex, race, and age groups.























.png)












No hay comentarios:
Publicar un comentario