
A new DRUG TRIALS SNAPSHOT is now available.
Drug Trials Snapshots: TURALIO
TURALIO is a drug used to treat adults with a tumor in the protective layer surrounding the tendons called Tenosynovial Giant Cell Tumor or TGCT. It should only be used in patients when the tumor limits daily activity, and if the tumor cannot be removed by surgery.
TURALIO is a capsule taken twice daily on an empty stomach.
See more Drug Trials Snapshots or contact us with questions at Snapshots@fda.hhs.gov.
TURALIO (pexidartinib)
(tur a' lee oh)
Daiichi Sankyo Inc.
Approval date: August 2, 2019
(tur a' lee oh)
Daiichi Sankyo Inc.
Approval date: August 2, 2019
DRUG TRIALS SNAPSHOT SUMMARY:
What is the drug for?
TURALIO is a drug used to treat adults with a tumor in the protective layer surrounding the tendons called Tenosynovial Giant Cell Tumor or TGCT. It should only be used in patients when the tumor limits daily activity, and if the tumor cannot be removed by surgery.
How is this drug used?
TURALIO is a capsule taken twice daily on an empty stomach.
What are the benefits of this drug?
Thirty-eight percent of 61 patients who were treated with TURALIO responded to the treatment with complete or partial shrinkage of their tumor. In comparison, none of the 59 patients who were treated with placebo had complete or partial shrinkage of their tumor.
Were there any differences in how well the drug worked in clinical trials among sex, race and age?
- Sex: TURALIO worked similarly in men and women.
- Race: The majority of patients were White. The number of patients of other races was limited; therefore, differences in how well TURALIO worked among races could not be determined.
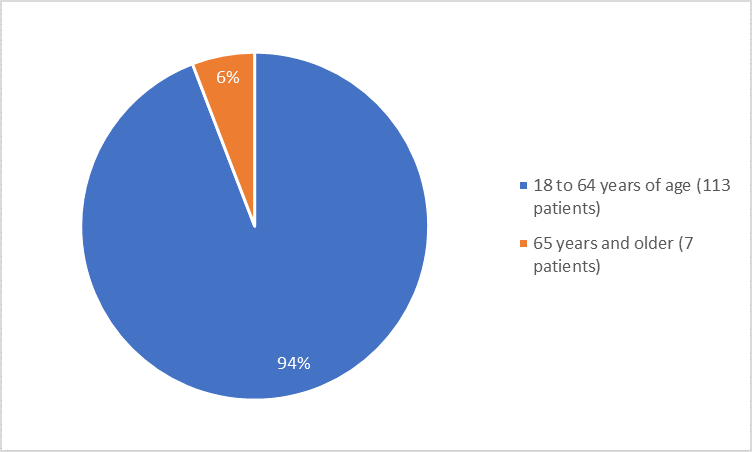
- Age: The majority of patients were adults younger than 65 years of age. The number of patients older than 65 years was limited; therefore, differences in how well TURALIO worked between patients younger and older than 65 years of age could not be determined.
What are the possible side effects?
TURALIO can cause serious liver injury which could lead to death. This is the reason that TURALIO is only available through a restricted program called Risk Evaluation and Mitigation Strategy (REMS) program.
TURALIO may cause harm to a fetus.
The most common side effects of TURALIO are abnormal liver tests, hair color changes, and tiredness.
Were there any differences in side effects among sex, race and age?
- Sex: The occurrence of side effects was similar between men and women.
- Race: The majority of patients were White. The number of patients of other races was limited; therefore, differences in the occurrence of side effects among races could not be determined.
- Age: Most patients were adults younger than 65 years of age. The number of patients older than 65 years of age was limited; therefore, differences in the occurrence of side effects between patients younger and older than 65 years of age could not be determined.
WHO WAS IN THE CLINICAL TRIALS?
Who participated in the clinical trials?
The FDA approved TURALIO based on evidence from 1 clinical trial (NCT02371369) of 120 patients with Tenosynovial Giant Cell Tumor or TGCT. The trial was conducted in Australia, Canada, Europe, and the United States.
Figure 1 summarizes how many men and women were in the clinical trial used to evaluate efficacy
Figure 1. Baseline Demographics by Sex
FDA Review
Figure 2 and Table 1 summarize the percentage of patients by race in the clinical trial.
Figure 2. Baseline Demographics by Race
FDA Review
Table 1. Demographics of Trials by Race
Race | Number of Patients | Percentage of Patients |
White | 106 | 88% |
Black or African-American | 4 | 3% |
Asian | 3 | 3% |
Native Hawaiian or Other Pacific Islander | 4 | 3% |
American Indian or Alaskan Native | 2 | 2% |
Other | 1 | 1% |
FDA Review
Figure 3 summarizes the percentage of patients by age group in the clinical trial.
Figure 3. Baseline Demographics by Age
FDA Review
How were the trials designed?
The benefits and side effects of TURALIO were evaluated in one clinical trial that enrolled adult patients with TGCT. All patients had limitations of daily activities due to the tumor and could not have the tumor removed by surgery. Patients received TURALIO or placebo twice a day until the disease worsened or they could not tolerate the side effects. Neither the patients nor the healthcare providers knew which treatment was being given for 24 weeks. After week 24, patients who received treatment with TURALIO could continue treatment and patients who received placebo could be treated with TURALIO. The benefit of TURALIO was evaluated at Week 25 by counting the patients who experienced partial or complete tumor shrinkage. The size of the tumor was evaluated by an imaging technique called magnetic resonance imaging (MRI).
GLOSSARY
CLINICAL TRIAL: Voluntary research studies conducted in people and designed to answer specific questions about the safety or effectiveness of drugs, vaccines, other therapies, or new ways of using existing treatments.
COMPARATOR: A previously available treatment or placebo used in clinical trials that is compared to the actual drug being tested.
EFFICACY: How well the drug achieves the desired response when it is taken as described in a controlled clinical setting, such as during a clinical trial.
PLACEBO: An inactive substance or “sugar pill” that looks the same as, and is given the same way as, an active drug or treatment being tested. The effects of the active drug or treatment are compared to the effects of the placebo.
SUBGROUP: A subset of the population studied in a clinical trial. Demographic subsets include sex, race, and age groups.
COMPARATOR: A previously available treatment or placebo used in clinical trials that is compared to the actual drug being tested.
EFFICACY: How well the drug achieves the desired response when it is taken as described in a controlled clinical setting, such as during a clinical trial.
PLACEBO: An inactive substance or “sugar pill” that looks the same as, and is given the same way as, an active drug or treatment being tested. The effects of the active drug or treatment are compared to the effects of the placebo.
SUBGROUP: A subset of the population studied in a clinical trial. Demographic subsets include sex, race, and age groups.




































No hay comentarios:
Publicar un comentario